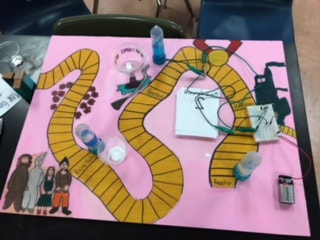This project was called Chemical dominoes and the goal was to create a board game that included chemical reactions. During this projects we had to include a phase change, creation of gas, single displacement reaction that lights up an LED light, and double displacement reaction. The board game could be simple but needed a level of difficulty and frustration. We had six weeks to finish our STEM project! We began by brainstorming a theme for our game and ideas of what reactions we wanted to use. After changing our ideas multiple times, we decided to use the theme The Wizard of Oz. During our project we conducted three reactions. During our first reaction we combined HCL and baking soda, our second we combined vinegar and baking soda, and our third we combined tin and NaHCO3.
Content:
Chemical Change: A change in the chemical properties or composition of a material
Physical Change: A change in the shape, phase, or form of an object in which it retains its identity (composition, chemical formula, etc.)
Chemical Reaction: A chemical change that happens when two or more substances react and change to form a new substance or substances.
Atom: the basic unit of a chemical element
Ion: Versions of an element that are not electrically neutral, meaning that they have a charge
Activity Series: The order of the reactivity of elements that allows us to predict whether a metal will displace the cation in a solution
Solubility Guidelines: Guidelines that allow us to predict whether two aqueous solutions will form a soluble solution or a precipitate
Proton: a stable subatomic particle occurring in all atomic nuclei, with a positive electric charge equal in magnitude to that of an electron, but of opposite sign
Neutron: a subatomic particle of about the same mass as a proton but without an electric charge, present in all atomic nuclei except those of ordinary hydrogen
Electron: a stable subatomic particle with a charge of negative electricity, found in all atoms and acting as the primary carrier of electricity in solids
Element: An element is a piece of matter that is made up of only one type of atom. An example would be iron, which is made up of only iron atoms.
Compound: a thing that is composed of two or more separate elements; a mixture
Single Replacement Reaction: chemical reaction where an element reacts with a compound and takes the place of another element in that compound
Double Displacement Reaction: where two compounds react, and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products
Synthesis Reaction: multiple reactants combine to form a single product
Decomposition Reaction: which a single compound breaks down into two or more elements or new compounds
Combustion: which something reacts with oxygen - commonly known as burning
Aqueous: of or containing water, typically as a solvent or medium
Precipitate: The precipitate forms because the solid is insoluble in water
Catalyst: A catalyst is a substance that speeds up a chemical reaction, but is not consumed by the reaction
Reflection:
This project was a huge success to me! We had our ups and downs but it turned out awesome in the end and that is all that matters. Our group worked well together and we all seemed to enjoy each other’s company. We were a very productive group, taking full advantage of our time in class. This group has taught me s ton! Two things I need to improve on is the effort I put into the group and leadership. This group would always point out when I needed to be helpful and pitch in a lot more and I really think that helps. I need to focus on staying in my group and on topic rather than lingering around with my friends in other groups. I want to be more engaged and willing because I realized it was disrespectful to my group members and not helping me learn when I was off task. I also want to work on my leadership skills because I need to improve them. Two things I thought I did very well with this group was throwing out new ideas and helping with the whole layout of our project. This project forced you to think outside the box which I loved. During this project I challenged myself to stay off my phone and I thought I did a really good job of that. This was a cool project because we had so much freedom and choices, I really enjoyed it. Overall I would love to do this project again in the future when I am more mature and can have different ideas!